Letter to the Editor: “The effect of abolishing instructions to fast prior to contrast-enhanced CT on the incidence of acute adverse reactions”
Authors: 1,2Shunji Mugikura, MD, PhD and 1Kei Takase, MD, PhD
Affiliations: 1Department of Diagnostic Radiology
Graduate School of Medicine, Tohoku University, 1-1
Seiryo-machi, Aoba-ku, Sendai, Japan 980-8574
2Division of Image Statistics
Tohoku medical megabank organization, Tohoku University, Seiryo-machi, Aoba-ku, Sendai, Japan 980-8574
Dear Editor in Chief,
We read the article entitled “The effect of abolishing instructions to fast prior to contrast-enhanced CT on the incidence of acute adverse reactions” written by Tsushima Y et al. with great interest [1].
We have some comments regarding the problems with generic iodine contrast agent pre-filled syringes for CT and injector compliance occurring in Japan. There is an increasing trend of using generic drugs to reduce medical costs. However, guaranteed compatibility between generic drugs (contrast agents with pre-filled syringes) and medical devices (injector) is vital for patients who underwent contrast-enhanced CT scans. The problem relating to the compatibility and warranty of generic pre-filled syringes and injectors is similar to the compatibility and warranty issues of smartphones and third-party power adapters.
In Japan, an iodine contrast agent is used in a pre-filled syringe for contrast-enhanced CT imaging. It is commonly administered using an injector at least once per scan. Indeed, as Tsushima Y et al. mentioned, one of five official or non-generic prefilled iodine contrast agents for CT was intravenously administered with an automatic injector (Dual Shot Nemoto Kyorindo, Tokyo, Japan). The automatic injector used in their study guaranteed the use of such five official or non-generic prefilled iodine contrast agents for CT without guaranteeing the use of generic prefilled iodine contrast agents.
Japanese national policy recommends the use of generic drugs to reduce medical costs. In contrast, when using medical devices, it is mandatory to comply with the usage method stipulated on the package insert by the manufacturer of the device in question [2]. However, to the best of our knowledge, the injectors sold in Japan are not guaranteed to be compatible with all generic pre-filled syringes.
Recently, in our institution, which is a large hospital in the Tohoku region of Japan, we deferred the decision of applying generic drugs because the injectors were not guaranteed to be compatible with all generic pre-filled syringes. According to the injector’s package insert, unspecified pre-filled syringes should not be used because flow rate accuracy and alarm function are not guaranteed and may also cause medical accidents [3]. A brief scheme could help getting the key point from this issue (Figure).
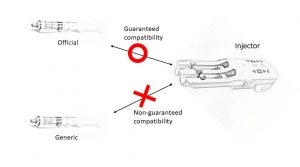
Figure 1. The automatic injector used in Japan guaranteed the use of official prefilled iodine contrast agents for CT without guaranteeing the use of generic prefilled iodine contrast agents.
The market share of generic pre-filled syringes in Japan is approximately 30% (unit), and continues to increase annually [4]. Given the rapid and widespread increase in using these generic pre-filled syringes, we believe that very few injectors, which do not currently guarantee compatibility, will cause possible problematic issues. The question of why these injector manufacturers have excluded guaranteed compatibility with generic pre-filled syringes may be related to the present situation in Japan, where one of the major injector manufacturers—which holds approximately 90% of the market share—is making various technological developments and product improvements in collaboration with the original product manufacturer [5]. Another problem involves the difficulty of checking and guaranteeing the compatibility of all generic pre-filled syringes that have become available on the market in rapid succession. The Japanese government should take this issue into account.
Disclaimer
While the authors are associated with the Japan Radiological Society, the views presented in this paper are the authors’ personal opinions, and not the society’s official view.
References
[1] Tsushima Y, Yuko Seki Y, Nakajima T, et al (2020) The effect of abolishing instructions to fast prior to contrast-enhanced CT on the incidence of acute adverse reactions. Insights Imaging. 11: 113.
[2] Ministry of Health, Labor and Welfare, Japan: I-Sei-Shi-Hatsu: 0330001, I-Sei-Ken-Hatsu: 0330018 http://hcea.umin.ac.jp/files/pdf/070330unyoutuuti.pdf 2007, 30th March.
[3] Package insert of Dual Shot GX7, January 23th, 2015 (6th edition). approved number 223ABBZX00051000
[4] National Database of Health Insurance Claims and Specific Health Checkups of Japan. Ministry of Health, Labor and Welfare. Japan
http://www.mhlw.go.jp/stf/seisakunitsuite/bunya/kenkou_iryou/iryouhoken/reseputo/index.html
[5] Next-generation medical service with Radio Frequency Identification (RFID) (in Japanese)ASCII. October 2007. Page 96
